Adhesion Barrier Market Summary
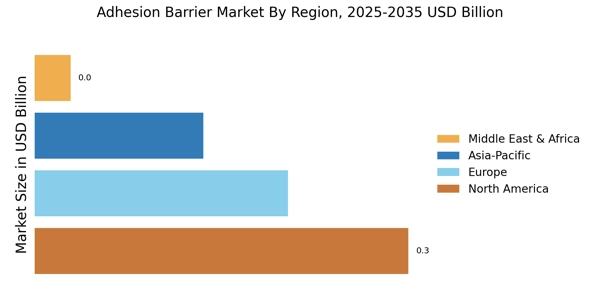
According to MRFR analysis, the Adhesion Barrier Market Size was valued at USD 0.6894 Billion in 2024. The market is projected to grow from USD 0.7328 Billion in 2025 to USD 1.349 Billion by 2035, registering a CAGR of 6.2% during the forecast 2025–2035. North America led the market with over 43.52% share, generating around USD 0.3 billion in revenue.
The Adhesion Barrier Market is expanding due to the increasing volume of surgical procedures, rising awareness of postoperative complications, and growing focus on improving patient outcomes. Key trends include adoption of advanced bioresorbable materials, increasing use in minimally invasive surgeries, and continuous innovation aimed at reducing adhesion-related complications and enhancing recovery.
Evidence shows that postoperative adhesions develop in up to 90% of patients undergoing abdominal surgery, making prevention strategies such as adhesion barriers increasingly important.
Key Market Trends & Highlights
The Adhesion Barrier Market is poised for substantial growth driven by technological advancements and increasing surgical procedures.
- The market experiences rising demand for minimally invasive procedures, particularly in North America.
- Innovations in material science are enhancing the efficacy of adhesion barriers, especially in abdominal surgery.
- Regulatory influences are shaping product development, with a notable focus on safety and effectiveness.
- Key drivers include the increasing number of surgical procedures and the growing awareness of postoperative complications.
Market Size & Forecast
| 2024 Market Size | 0.6894 (USD Billion) |
| 2035 Market Size | 1.349 (USD Billion) |
| CAGR (2025 - 2035) | 6.29% |
Major Players
Companies such as Baxter International(US), Johnson & Johnson (US), Medtronic(US), C.R. Bard (US), FzioMed (US), Hollister Incorporated (US), Sanofi (FR), Smith & Nephew (GB), Stryker Corporation (US) are some of the major participants in the Global Adhesion Barrier Market.