
細胞治療市場
細胞治療市場 概要
MRFRの分析によると、細胞治療市場の規模は2024年に161.4億米ドルと推定されています。細胞治療業界は、2025年に175.3億米ドルから2035年には399.2億米ドルに成長すると予測されており、2025年から2035年の予測期間中に年平均成長率(CAGR)は8.58を示します。
主要な市場動向とハイライト
細胞治療市場は、技術の進歩と治療法の受け入れの増加により、堅調な成長を遂げています。
- 北米は、研究開発への大規模な投資によって、細胞治療の最大の市場であり続けています。アジア太平洋地域は、医療費の増加と患者人口の増加により、最も成長している市場として浮上しています。腫瘍学は最大のセグメントとして市場を支配し続けており、神経障害は最も成長しているセグメントとして急速に注目を集めています。市場の主要な推進要因には、慢性疾患の増加と細胞処理技術の進歩が含まれ、治療オプションが向上しています。
市場規模と予測
| 2024 Market Size | 16.14 (米ドル十億) |
| 2035 Market Size | 39.92 (米ドル十億) |
| CAGR (2025 - 2035) | 8.58% |
主要なプレーヤー
ノバルティス (CH)、ギリアド・サイエンシズ (US)、ブリストル・マイヤーズ スクイブ (US)、アムジェン (US)、セルジーン (US)、サンガモ・セラピューティクス (US)、ブルーバード・バイオ (US)、セリヤド (BE)、カイト・ファーマ (US)、フェイト・セラピューティクス (US)
細胞治療市場 トレンド
細胞治療市場は、技術の急速な進歩と細胞メカニズムの理解の向上によって特徴づけられる変革の段階を迎えています。この進展は、慢性疾患や遺伝性疾患の増加に大きく起因しており、革新的な治療法が求められています。研究者たちが幹細胞やその他の細胞治療の可能性を深く掘り下げる中、市場は大きな成長の準備が整っているようです。さらに、規制当局はこれらの変化に適応し、新しい治療法の導入を促進するために承認プロセスを簡素化しています。この規制機関の柔軟な対応は、細胞治療市場の全体的な状況を改善し、革新を促進する環境を育む可能性があります。技術の進歩に加えて、学術機関、バイオテクノロジー企業、製薬会社間の協力がますます一般的になっています。このようなパートナーシップは、研究開発の努力を加速させ、未解決の医療ニーズに応える新しい治療法の出現につながる可能性があります。さらに、細胞ベースの治療法への投資の増加は、利害関係者からの強い関心を示しており、市場のダイナミクスをさらに刺激するかもしれません。細胞治療市場が進化し続ける中、これらのトレンドを注意深く監視することが重要であり、将来の発展に大きな影響を与える可能性があります。
技術の進歩
細胞治療市場は、治療の有効性と安全性を高める驚くべき技術革新を目の当たりにしています。遺伝子編集や個別化医療などの技術がますます普及しており、特定の患者のニーズに応じた治療法が可能になっています。この傾向は、より正確で効果的な治療オプションへのシフトを示しています。
共同研究イニシアチブ
細胞治療市場におけるさまざまな利害関係者間の共同努力が顕著に増加しています。学術機関と業界のプレーヤー間のパートナーシップは、知識とリソースの共有の環境を育んでおり、新しい治療法の開発を加速させる可能性があります。この協力は、分野の既存の課題を克服するために重要であるようです。
規制の進化
細胞治療市場を取り巻く規制の状況は、技術の急速な進歩に対応して進化しています。規制機関は承認プロセスを簡素化しており、革新的な治療法への迅速なアクセスを促進する可能性があります。この進化は、現代医療における細胞ベースの治療の重要性が高まっていることを示唆しています。
細胞治療市場 運転手
慢性疾患の増加
慢性疾患、例えば癌、糖尿病、心血管障害の発生率の増加は、細胞治療市場の主要な推進要因です。これらの疾患がますます一般的になるにつれて、革新的な治療オプションへの需要が高まります。再生医療と標的治療の可能性を提供する細胞治療は、医療提供者や患者の間で注目を集めています。最近の推定によれば、慢性疾患の世界的な負担は前例のないレベルに達することが予想されており、高度な治療ソリューションが必要とされています。この傾向は、慢性疾患に関連する増大する医療課題に対処しようとする利害関係者による細胞治療市場への研究開発への投資を促進する可能性があります。
細胞処理技術の進展
細胞処理および製造における技術革新は、細胞治療市場に大きな影響を与えています。細胞の分離、増殖、修飾のための高度な技術が、細胞治療の効率と有効性を向上させています。たとえば、自動化された細胞処理システムは、生産を効率化し、コストを削減し、汚染リスクを最小限に抑えています。細胞処理技術の市場は大幅に成長することが予測されており、今後数年間で20%以上の年平均成長率が見込まれています。これらの進展は、新しい治療法の開発を促進するだけでなく、既存の治療法のスケーラビリティを向上させ、細胞治療市場の範囲を拡大しています。
再生医療への投資の増加
再生医療に向けた投資の急増は、細胞治療市場の顕著な推進要因です。ベンチャーキャピタル企業や製薬会社は、細胞治療が治療のパラダイムを革新する可能性をますます認識しています。報告によると、再生医療スタートアップへの資金提供は数十億ドルに達しており、革新的な治療ソリューションへの強い関心を反映しています。この資本の流入は、細胞治療市場における研究イニシアチブ、臨床試験、製品開発を加速させる可能性があります。より多くの治療法が規制当局の承認を受けるにつれて、市場は拡大し、医療分野の利害関係者に新たな機会を提供することが期待されています。
規制支援と効率的な承認プロセス
規制当局は、細胞治療の開発と商業化を支援するための取り組みを強化しており、これは細胞治療市場の重要な推進要因です。革新的な治療法の承認プロセスを簡素化することを目的とした取り組みが実施されており、命を救う治療法への迅速なアクセスが可能になっています。たとえば、再生医療製品のための迅速な承認経路の導入は、企業が細胞治療の研究に投資することを促しています。この規制環境は、革新を促進するだけでなく、市場の信頼を高め、利害関係者が新しい治療法を市場に投入するためのより好ましい環境を認識しています。規制の枠組みが進化し続ける中、細胞治療市場は大きな成長が期待されています。
細胞療法に対する認識と受容の高まり
細胞治療に対する公衆の認識と受容は着実に高まっており、細胞治療市場の成長に寄与しています。教育キャンペーンや成功事例がこれらの先進的な治療法を解明し、より情報に基づいた患者集団を育成しています。患者が細胞治療の潜在的な利点をより認識するにつれて、需要は高まると考えられます。さらに、医療提供者はこれらの革新的な解決策をますます支持しており、患者の治療結果を改善する可能性を認識しています。この認識の変化は市場の成長を促進すると予想されており、より多くの個人が自らの医療条件に対する細胞治療の選択肢を求めるようになるでしょう。
市場セグメントの洞察
用途別:腫瘍学(最大)対神経障害(最も成長が早い)
細胞治療市場において、腫瘍学は最大のセグメントであり、世界的な癌の発生率の上昇と革新的な治療アプローチの受け入れの高まりにより、重要な市場シェアを占めています。CAR-Tや腫瘍浸潤リンパ球療法を含む腫瘍学療法は、治療の風景において重要なプレーヤーとして確立されており、この分野での大規模な投資と進展を促進しています。一方、神経障害は新興セグメントとして注目を集めており、アルツハイマー病やパーキンソン病などの状態に対する細胞治療の可能性が研究によって明らかになるにつれて急速に成長しています。この変化は、バイオ医薬品企業がこれらの慢性で衰弱させる状態に対する新しい介入の開発にますます注力していることから明らかであり、患者に新たな希望を提供しています。
腫瘍学(主導)対心血管疾患(新興)
腫瘍学の細胞療法はその優位性を確保し、市場の革新の最前線に立っています。CAR-Tや幹細胞移植などのさまざまな治療法が実質的な臨床効果を示しています。これらの療法はしばしば個別化されており、腫瘍医や患者に共鳴するカスタマイズされた治療オプションを提供します。一方、心血管疾患は再生医療アプローチが注目されている新興セグメントです。心筋梗塞後の損傷した組織を修復したり、心不全を治療するために幹細胞や工学的心筋組織を活用することへの関心が高まっており、このセグメントは注目を集めています。臨床試験が進行し、成功事例が出てくるにつれて、心血管疾患セグメントは細胞療法市場における魅力的な成長分野となるでしょう。
タイプ別:自家細胞療法市場(最大)対同種細胞療法市場(最も成長が早い)
細胞治療市場は、自己細胞治療市場、同種細胞治療市場、幹細胞治療市場の3つの主要なセグメントで構成されています。これらの中で、自己細胞治療市場は、患者自身の体から細胞を取り出すパーソナライズされたアプローチにより、最大の市場シェアを占めています。同種細胞治療市場は、オフ・ザ・シェルフソリューションの可能性により急速に拡大しているセグメントとして続いており、より広範な患者層への治療のアクセスを向上させています。幹細胞治療市場は影響力がありますが、現在は市場の中で小さなセグメントを占めており、さまざまな革新的な治療において重要な役割を果たしています。
自家細胞治療市場(主導)対同種細胞治療市場(新興)
自己細胞療法市場は、個々の患者から細胞を抽出し、再導入する前に操作する患者特有の方法論によって特徴付けられ、個別化された治療計画を導きます。このセグメントは、強力な規制の支援と確立されたプロトコルにより安定した市場ポジションを享受しており、提供者と患者の両方に信頼を育んでいます。それに対して、同種細胞療法市場は業界の重要なプレーヤーとして浮上し、オフ・ザ・シェルフの治療法を提供することで、患者のアクセスを向上させ、待機時間を短縮しています。より便利で即使用可能な治療製品へのシフトが、このセグメントへの投資と成長を促進しており、細胞療法における革新の魅力的な分野となっています。
配達方法による:静脈内注射(最大)対皮下注射(最も成長が早い)
細胞治療市場におけるデリバリーメソッドセグメントは、主に静脈注射によって特徴付けられています。これは、細胞ベースの治療法を直接血流に届ける確立された有効性により、最大の市場シェアを保持しています。皮下注射は、現在のシェアは小さいものの、注射技術の進歩に伴い、増加する市場セグメントを獲得する可能性が大きいです。筋肉内注射および皮内注射は、より小さな市場の足跡を持っていますが、特定の治療用途において独自の利点を提供し、業界における多様なデリバリーメソッドに寄与しています。デリバリーメソッドセグメント内の成長トレンドは好ましく、特に皮下注射は、投与の容易さと患者への副作用の軽減により、注目を集めています。針技術や製剤の革新は、皮下注射および皮内注射の成長を促進すると予測されており、静脈注射はその信頼性から依然として主流です。患者中心の治療アプローチの進化は、よりアクセスしやすいデリバリーメソッドの必要性をさらに強調しており、市場を前進させています。
静脈注射(主流)対皮下注射(新興)
静脈注射は、治療薬を迅速に全身循環に高濃度で投与する効果的な方法であるため、細胞治療市場において支配的な投与方法として残っています。この方法は、予測可能な薬物動態と確立された安全性プロファイルを持つため、複雑な治療に適しています。一方、皮下注射は自己投与が可能であり、患者の遵守が良好であるため、好まれる代替手段として浮上しています。製剤技術の進展に伴い、皮下注射は医療コストの削減や患者の利便性の向上といった利点を提供することが期待されています。これら二つの方法の比較は、治療効果を維持しながら、さまざまな患者のニーズに対応する業界のシフトを示しています。治療的な効果を維持しながら。
エンドユーザーによる:病院(最大)対研究機関(最も成長している)
細胞治療市場において、病院は最大のセグメントを占めており、患者治療のためにこれらの先進的な治療法を広く利用しています。彼らの優位な地位は、慢性疾患の増加と細胞ベースの治療法の受け入れの高まりによって支えられています。研究機関は市場シェアは小さいものの、この分野における革新と開発において重要な役割を果たしているため、急速に注目を集めており、市場で最も成長しているセグメントとなっています。
病院(支配的)対研究機関(新興)
病院は、細胞治療市場において、これらの治療法の主要な提供ポイントとして重要な役割を果たしており、入院および外来サービスの両方を提供しています。彼らは、複雑な治療を行うための最先端の技術と専門チームを備えており、確立されたインフラと患者へのアクセスにより、支配的なセグメントとなっています。一方、科学的発見へのコミットメントで知られる研究機関は、細胞治療における革新の温床として機能しています。彼らは新しい治療法の開発や臨床試験の実施を通じて市場における新たな機会に大きく貢献しており、研究努力を進めるための資金やパートナーシップを引き寄せています。
地域の洞察
北米 : イノベーションと投資のリーダー
北米は細胞治療の最大の市場であり、世界市場シェアの約45%を占めています。この地域は、研究開発への強力な投資、堅固な規制枠組み、先進的な治療法への需要の高まりから恩恵を受けています。FDAの細胞治療に対する支持的な姿勢は、イノベーションを促進し、治療オプションの急速な進展をもたらしました。慢性疾患の増加と高齢化する人口は、市場の成長をさらに促進しています。アメリカ合衆国はこの市場の主要な貢献者であり、ノバルティス、ギリアド・サイエンシズ、ブリストル・マイヤーズ スクイブなどの主要企業が先頭を切っています。競争環境は、企業がポートフォリオを強化しようとする中で、重要な合併や買収によって特徴づけられています。カナダも重要な役割を果たしており、バイオテクノロジーへの投資が増加し、革新的な治療法の開発に焦点を当てているため、北米は細胞治療の進展の中心地となっています。
ヨーロッパ : 新興の規制枠組み
ヨーロッパは細胞治療の第二の市場であり、世界市場シェアの約30%を保持しています。この地域では、医療への投資の増加とバイオテクノロジーの進展により、革新的な治療法への需要が急増しています。欧州医薬品庁(EMA)などの規制機関は、承認プロセスの簡素化に積極的に取り組んでおり、これにより市場の成長がさらに促進されると期待されています。遺伝性疾患や癌の増加も需要の重要な推進要因です。この地域の主要国にはドイツ、イギリス、フランスがあり、セリヤドやカイト・ファーマなどの主要企業が多数存在しています。競争環境は進化しており、バイオテクノロジー企業と研究機関の間のコラボレーションに焦点が当てられています。強力な学術研究の存在と好意的な規制環境が、この地域の細胞治療開発の魅力を高めています。EMAは「先進的な治療法の開発は、ヨーロッパにおける未解決の医療ニーズに対処するために重要である」と述べています。
アジア太平洋 : 急成長する市場の可能性
アジア太平洋地域は、細胞治療市場において急速に重要なプレーヤーとして浮上しており、世界市場シェアの約20%を占めています。この地域は、医療インフラへの投資の増加、患者人口の増加、先進的な治療法への認識の高まりが特徴です。中国や日本などの国々が先頭を切っており、好意的な政府政策とバイオテクノロジーのイノベーションに焦点を当てています。個別化医療への需要もこの地域の市場成長を促進しています。中国は研究開発への大規模な投資を行っており、日本は先進的な医療システムで知られています。競争環境はますますダイナミックになっており、地元企業と国際企業が市場シェアを争っています。アムジェンやフェイト・セラピューティクスなどの主要企業が存在感を拡大し、この地域の細胞治療ソリューションの成長に寄与しています。アジア太平洋市場は、技術の進展と医療支出の増加により、今後数年間で大幅な拡大が見込まれています。
中東およびアフリカ : 未開拓の市場機会
中東およびアフリカ地域は、現在細胞治療の最小の市場であり、世界市場シェアの約5%を占めています。しかし、医療とバイオテクノロジーへの投資の増加により、重要な成長の可能性を秘めています。慢性疾患の増加と先進的な治療オプションの必要性が需要を促進しています。この地域の政府はバイオテクノロジーの重要性を認識し始めており、支持的な政策や資金イニシアティブが生まれています。南アフリカやUAEなどの国々が細胞治療の分野で重要なプレーヤーとして浮上しており、地元企業と国際企業の両方からの関心が高まっています。競争環境はまだ発展途上ですが、細胞治療研究を進めるためのコラボレーションやパートナーシップが顕著に増加しています。この地域が医療インフラへの投資を続ける中で、細胞治療の成長の可能性は大きく、投資家やイノベーターにとって注目のエリアとなっています。
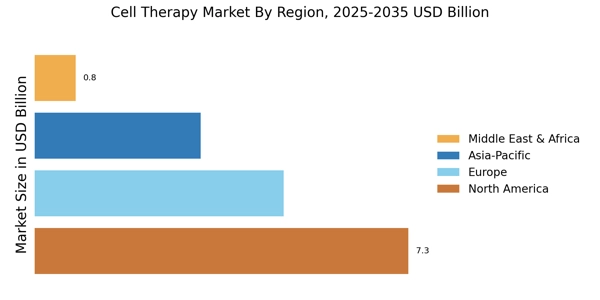
主要企業と競争の洞察
細胞治療市場は、現在、技術の急速な進展と革新的な治療オプションに対する需要の高まりによって推進される動的な競争環境を特徴としています。ノバルティス(スイス)、ギリアド・サイエンシズ(米国)、ブリストル・マイヤーズ スクイブ(米国)などの主要企業が最前線に立ち、それぞれが市場ポジションを強化するための独自の戦略を採用しています。ノバルティス(スイス)は、特にCAR-T細胞療法の分野において、戦略的パートナーシップやコラボレーションを通じてポートフォリオの拡大に注力しています。ギリアド・サイエンシズ(米国)は、特に血液悪性腫瘍のための次世代治療法の開発において、製品提供の革新を強調しています。ブリストル・マイヤーズ スクイブ(米国)は、細胞治療における能力を強化するために合併や買収を積極的に追求しており、競争力を高めています。これらの戦略は、革新を促進し、市場成長を推進する堅牢な競争環境に寄与しています。
ビジネス戦略に関して、企業は供給チェーンの効率を高め、コストを削減するために製造のローカライズを進めています。このアプローチは、オペレーションを効率化するだけでなく、市場の需要に迅速に対応することを可能にします。細胞治療市場は、いくつかの主要企業が重要な影響を及ぼしている中で、適度に分散しているようです。これらの企業の集合的な行動は、市場構造を形成し、革新と戦略的ポジショニングを通じてリーダーシップを争っています。
2025年8月、ノバルティス(スイス)は、固形腫瘍の治療を目的とした新しいCAR-T療法を共同開発するために、主要なバイオテクノロジー企業との画期的なパートナーシップを発表しました。このコラボレーションは、困難な腫瘍学の分野に対処するノバルティスの能力を強化することが期待されており、同社をこのニッチのリーダーとして位置づける可能性があります。このパートナーシップの戦略的重要性は、開発タイムラインを加速し、CAR-T技術の治療適用範囲を広げる可能性にあります。
2025年9月、ギリアド・サイエンシズ(米国)は、細胞治療の生産に特化した新しい製造施設を発表し、これにより生産能力が大幅に向上することが期待されています。この投資は、革新的な治療法に対する高まる需要に応えるというギリアドのコミットメントを強調しており、供給チェーンの信頼性を高めるための戦略的な動きを反映しています。製造能力を強化することで、ギリアドは医療提供者や患者への製品のタイムリーな提供を確保し、市場ポジションを強化することを目指しています。
2025年10月、ブリストル・マイヤーズ スクイブ(米国)は、遺伝子編集技術に特化した小規模なバイオテクノロジー企業の買収を完了しました。この買収は、特に細胞および遺伝子治療の分野におけるブリストル・マイヤーズ スクイブの研究開発能力を強化する可能性があります。この動きの戦略的重要性は、同社の製品パイプラインを多様化し、次世代治療法の開発を加速する可能性にあります。これにより、市場における競争力が強化されるでしょう。
2025年10月現在、細胞治療市場は、デジタル化、持続可能性、治療開発における人工知能の統合を強調するトレンドを目撃しています。戦略的アライアンスは、企業が革新を推進するためのコラボレーションの価値を認識する中で、競争環境をますます形成しています。今後、競争の差別化は進化することが予想され、価格競争から革新、技術の進展、供給チェーンの信頼性に焦点を当てたものへと明確にシフトするでしょう。この移行は、企業が市場でのポジショニングを再定義する可能性があり、最終的には患者に対してより良い治療オプションを提供することにつながるでしょう。
細胞治療市場市場の主要企業には以下が含まれます






業界の動向
細胞治療市場の最近の動向は、慢性疾患や癌に対する革新的な治療法への関心が高まっていることを示しています。ブルーバード・バイオ、アダプティミューン、ギリアド・サイエンシズなどの著名な企業は、先進的な細胞技術を活用するために研究開発の取り組みを拡大することに注力しています。
2023年10月、アダプティミューンは固形腫瘍を対象としたT細胞療法の試験から有望な初期結果を報告し、セクターへの投資家の関心を高めました。合併と買収の観点では、ギリアド・サイエンシズの子会社であるカイト・ファーマが、2023年9月にTCR2セラピューティクスの買収を発表し、細胞治療ポートフォリオの強化を目指しています。
細胞治療の市場評価は、規制機関からの承認率の増加と個別化医療の受容の高まりにより、新たな高みに達することが予測されています。アストラゼネカのCAR-T療法の進展も、市場のダイナミクスを変える要因となっています。
過去数年間、細胞治療における技術の統合は大きな進展を遂げており、ノバルティスは新しいT細胞療法を商業化に向けてリードしています。これらの開発は、医療分野における効果的な治療ソリューションの需要の高まりに応えるために調整された堅実な上昇軌道を示しています。
今後の見通し
細胞治療市場 今後の見通し
細胞治療市場は、2024年から2035年までの間に8.58%のCAGRで成長すると予測されており、これは技術の進歩、投資の増加、個別化医療の需要の高まりによって推進されます。
新しい機会は以下にあります:
- 効率を向上させるための自動細胞処理システムの開発。
- 特化した細胞治療ソリューションで新興市場への拡大。
- 革新的な製品パイプラインのためのバイオテクノロジー企業との戦略的パートナーシップ。
2035年までに、細胞治療市場は堅調であり、 substantial growth and innovationを反映することが期待されています。
市場セグメンテーション
細胞治療市場の提供方法の展望
- 静脈注射
- 皮下注射
- 皮内注射
- 筋肉注射
細胞療法市場のタイプの見通し
- 自己細胞療法
- 同種細胞療法
- 幹細胞療法
細胞治療市場のアプリケーション展望
- 腫瘍学
- 心血管疾患
- 神経障害
- 整形外科疾患
細胞治療市場のエンドユーザーの展望
- 病院
- 研究機関
- 外来手術センター
レポートの範囲
| 市場規模 2024 | 16.14(億米ドル) |
| 市場規模 2025 | 17.53(億米ドル) |
| 市場規模 2035 | 39.92(億米ドル) |
| 年平均成長率 (CAGR) | 8.58% (2024 - 2035) |
| レポートの範囲 | 収益予測、競争環境、成長要因、トレンド |
| 基準年 | 2024 |
| 市場予測期間 | 2025 - 2035 |
| 過去データ | 2019 - 2024 |
| 市場予測単位 | 億米ドル |
| 主要企業プロファイル | 市場分析進行中 |
| カバーされるセグメント | 市場セグメンテーション分析進行中 |
| 主要市場機会 | 個別化医療の進展が細胞治療市場の成長を促進します。 |
| 主要市場ダイナミクス | 革新的な治療法に対する需要の高まりが、細胞治療の環境における競争力と規制の適応を促進します。 |
| カバーされる国 | 北米、ヨーロッパ、アジア太平洋、南米、中東・アフリカ |
市場のハイライト
FAQs
2024年の時点での細胞治療市場の現在の評価額はどのくらいですか?
2035年の細胞治療市場の予測市場規模はどのくらいですか?
2025年から2035年までの細胞治療市場の予想CAGRはどのくらいですか?
細胞治療市場で最も高い成長が期待されるアプリケーションセグメントはどれですか?
自家細胞療法の市場は、同種細胞療法とどのように比較されますか?
細胞治療市場で支配的になると予想される配送方法は何ですか?
細胞治療市場で最も成長が見込まれるエンドユーザーセグメントはどれですか?
細胞治療市場の主要なプレーヤーは誰ですか?
細胞治療市場における神経障害セグメントの予測成長率はどのくらいですか?
幹細胞治療の市場は、他のタイプの細胞治療とどのように比較されますか?
Research Approach
Secondary Research
The secondary research process involved comprehensive analysis of regulatory databases, peer-reviewed medical journals, clinical trial repositories, and authoritative health organizations. Key sources included the US Food & Drug Administration (FDA) Center for Biologics Evaluation and Research (CBER), European Medicines Agency (EMA) Committee for Advanced Therapies (CAT), Pharmaceuticals and Medical Devices Agency (PMDA) Japan, National Medical Products Administration (NMPA) China, Health Canada, International Society for Cell & Gene Therapy (ISCT), American Society of Gene and Cell Therapy (ASGCT), Alliance for Regenerative Medicine (ARM), Foundation for Accreditation of Cellular Therapy (FACT), Joint Accreditation Committee-ISCT & EBMT (JACIE), ClinicalTrials.gov, National Institutes of Health (NIH) SEED Program, National Center for Biotechnology Information (NCBI/PubMed), World Health Organization (WHO) International Classification of Diseases, EU Clinical Trials Register, and national advanced therapy medicinal product (ATMP) registries from key markets. These sources were used to collect clinical trial statistics, regulatory approval data (RMAT/ATMP designations), manufacturing capacity analyses, patient treatment volumes, and competitive landscape intelligence for autologous cell therapies (CAR-T, TCR-T), allogeneic cell therapies (NK cells, iPSC-derived), and stem cell therapies (MSC, HSC).
Primary Research
To gather both qualitative and quantitative insights, supply-side and demand-side stakeholders were interviewed during the primary research process. Regulatory affairs professionals in ATMPs, CEOs, Chief Scientific Officers, Heads of Cell Therapy Manufacturing, and commercial directors from cell therapy companies, CDMOs (Contract Development and Manufacturing Organizations), and vector manufacturers were examples of supply-side sources. Principal investigators in Phase II/III cell therapy trials, hematologists-oncologists with expertise in administering CAR-T, medical directors of transplant centers, procurement leads for cell therapy treatment centers, and pharmacy directors from NCI-Designated Cancer Centers, academic medical centers, and specialized cellular therapy treatment facilities were among the demand-side sources. Primary research confirmed clinical pipeline timelines (Phase I through commercial), validated market segmentation across therapy types, and collected information on issues related to manufacturing scale-up, vector supply constraints, treatment center capacity expansion, outcomes-based contract pricing and reimbursement, and the competitive dynamics between allogeneic off-the-shelf platforms and autologous personalized therapies.
Primary Respondent Breakdown:
By Designation: C-level Primaries (40%), Director Level (35%), Others (25%)
By Region: North America (38%), Europe (30%), Asia-Pacific (25%), Rest of World (7%)
Market Size Estimation
Global market valuation was derived through revenue mapping and treatment volume analysis across CAR-T cell therapies, tumor-infiltrating lymphocytes (TIL), stem cell therapies, and emerging allogeneic platforms. The methodology included:
Identification of 50+ key manufacturers across North America, Europe, Asia-Pacific, and emerging markets, spanning large pharmaceutical companies, emerging biotechs, and specialized CDMOs
Product mapping across autologous cell therapies (patient-specific CAR-T, TCR-T), allogeneic cell therapies (donor-derived NK cells, iPSC-derived products), and stem cell therapies (mesenchymal stromal cells, hematopoietic stem cells, cord blood products)
Analysis of reported and modeled annual revenues specific to cell therapy portfolios, including approved therapies (Kymriah, Yescarta, Tecartus, Breyanzi, Abecma, Carvykti) and pipeline-stage products
Coverage of manufacturers and treatment centers representing 65-70% of global market share and patient volumes in 2024
Extrapolation using bottom-up (treatment-eligible patient volumes × therapy pricing by country/region, accounting for manufacturing success rates and vein-to-vein time) and top-down (manufacturer revenue validation, service provider capacity analysis) approaches to derive segment-specific valuations across oncology, cardiovascular, neurological, and orthopedic applications
FDA CBER Cellular & Gene Therapy Guidances
EMA Committee for Advanced Therapies (CAT) Scientific Recommendations
Alliance for Regenerative Medicine Sector Snapshots
ISCT Global (International Society for Cell & Gene Therapy) & Cytotherapy Journal
ASGCT (American Society of Gene & Cell Therapy)
NIH Regulatory Knowledge Guide for Cell and Gene Therapies
このレポートの無料サンプルを受け取るには、以下のフォームにご記入ください
Customer Stories

“This is really good guys. Excellent work on a tight deadline. I will continue to use you going forward and recommend you to others. Nice job”

“Thanks. It’s been a pleasure working with you, please use me as reference with any other Intel employees.”

“Thanks for sending the report it gives us a good global view of the Betaïne market.”

“Thank you, this will be very helpful for OQS.”

“We found the report very insightful! we found your research firm very helpful. I'm sending this email to secure our future business.”

“I am very pleased with how market segments have been defined in a relevant way for my purposes (such as "Portable Freezers & refrigerators" and "last-mile"). In general the report is well structured. Thanks very much for your efforts.”

“I have been reading the first document or the study, ,the Global HVAC and FP market report 2021 till 2026. Must say, good info! I have not gone in depth at all parts, but got a good indication of the data inside!”

“We got the report in time, we really thank you for your support in this process. I also thank to all of your team as they did a great job.”




