Global market valuation was derived through revenue mapping, device shipment analysis, and test strip volume modeling. The methodology included:
Identification of 50+ key manufacturers across North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa
Product mapping across self-monitoring blood glucose devices, continuous glucose monitoring systems ( sensors, transmitters, receivers), blood glucose test strips, and lancets
Technology segmentation analysis across electrochemical biosensors, optical glucose sensing, and non-invasive transdermal monitoring platforms
Analysis of reported and modeled annual revenues specific to diabetes monitoring portfolios, including recurring consumables revenue (test strips, lancets, CGM sensors) versus capital equipment sales
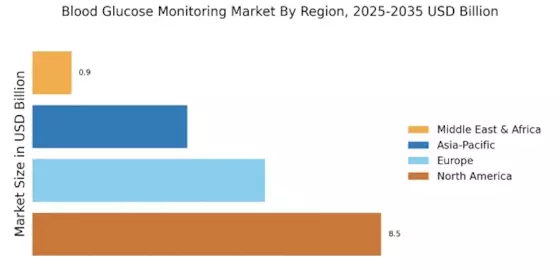
Coverage of manufacturers representing 75-80% of global market share in 2024
Extrapolation using bottom-up (diabetes prevalence × diagnosis rate × monitoring frequency × ASP by country/region) and top-down (manufacturer revenue validation, distributor margin analysis) approaches to derive segment-specific valuations for diabetes management, obesity management, and gestational diabetes monitoring applications across hospital, homecare, and ambulatory surgical center end-user settings
This methodology specifically incorporates diabetes-focused regulatory pathways (FDA 510k for Class II medical devices, ISO 15197 accuracy standards), diabetes epidemiology data sources (IDF Atlas), and BGM-specific stakeholder interviews (endocrinologists vs. dermatologists, diabetes educators vs. aesthetic practitioners) while maintaining the structural format you requested.