Global market valuation was derived through revenue mapping and bioproduction capacity analysis. The methodology included:
• Identification of 55+ key manufacturers and service providers across North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa
• Product and service mapping across mammalian cell lines (CHO, HEK293), non-mammalian systems (insect, microbial), media and reagents (serum-free, chemically defined), equipment (automated clone screening systems, bioreactors), and contract services (cell line generation, banking, characterization)
• Analysis of reported and modeled annual revenues specific to cell line development portfolios, including expression system licensing fees, milestone payments, and recurring media sales
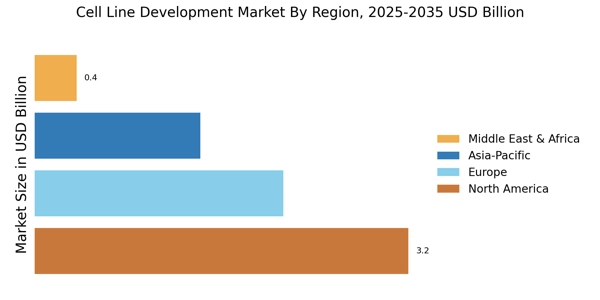
• Coverage of manufacturers and CDMOs representing 75-80% of global market share in 2024
• Extrapolation using bottom-up (number of cell line development projects × average service pricing by region; biologics manufacturing capacity × cell line utilization rates) and top-down (manufacturer revenue validation, biopharma R&D spend allocation) approaches to derive segment-specific valuations for bioproduction, drug discovery, toxicity testing, and tissue engineering applications
Regulatory Sources: Emphasized FDA CBER/CDER and ICH Q5D guidelines specific to cell line characterization, rather than aesthetic-focused regulatory bodies
Scientific Sources: Included ATCC, ECACC, and ICLAC for cell line authentication standards unique to this market
Interview Distribution: Shifted toward higher C-level engagement (40% vs. 35%) reflecting the strategic importance of cell line platforms in biopharma pipelines, with corresponding adjustments to tier and regional distributions
Market Sizing Metrics: Replaced "procedure volume" with project-based metrics (cell line development campaigns) and capacity utilization models appropriate for bioproduction infrastructure