末梢動脈疾患市場 概要
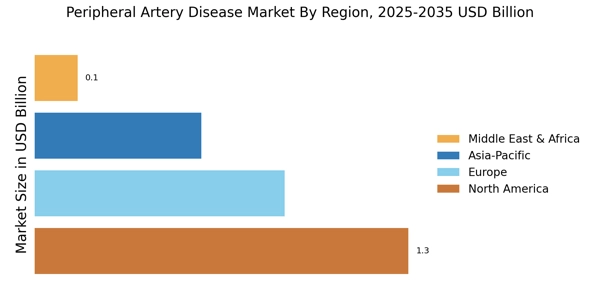
世界の末梢動脈疾患市場規模は2024年に28億9,900万米ドルと評価され、市場は2025年の30億9,600万米ドルから2035年までに59億7,900万米ドルに成長すると予測されており、2025年から2035年の予測期間中に6.8%のCAGRを記録します。北米は 44.84% 以上のシェアで市場をリードし、約 13 億米ドルの収益を生み出しました。
糖尿病、肥満、高血圧、高齢化率の増加により末梢動脈疾患の有病率が上昇しており、依然として市場の主な成長要因となっています。認識の向上、診断率の向上、低侵襲治療に対する需要により、世界的に導入が加速しています。
WHO によると、心血管疾患により世界中で年間約 1,790 万人が死亡しており、これは全世界の死亡者数のほぼ 32% に相当します。末梢動脈疾患は全身性アテローム性動脈硬化症の主要な症状であるため、心血管疾患の負担の増大が PAD の診断および治療ソリューションの需要を支え続けています。
主要な市場動向とハイライト
末梢動脈疾患市場は、技術の進歩と人口動態の変化によって大幅な成長が見込まれています。
- 北米は2024年に世界収益の44.84%以上を占め、末梢動脈疾患市場をリードしました。
- ヨーロッパの末梢動脈疾患市場は2024年に8億6,970万米ドルに達し、世界市場シェアの約30%を占めます。
- 低侵襲血管処置の採用増加により、デバイスは 2024 年に約 47% のシェアを獲得して市場を独占しました。
- 脂質低下薬は、2024 年には医薬品セグメントの約 44% を占め、長期的な心血管リスク管理をサポートしています。
- 病院と診療所は、高度な血管治療機能とインフラストラクチャに支えられ、2024 年には約 63% の市場シェアを獲得しました。
- 北米は、心血管疾患治療の旺盛な需要に支えられ、2024年に約13億米ドルの収益を上げました。
市場規模と予測
| 2024年の市場規模 | 2.899 (USD Billion) |
| 2035年の市場規模 | 5.979 (USD Billion) |
| CAGR (2025 - 2035) | 6.8% |
| 2024 年に最大の地域市場シェアを獲得 | 北米 |
主要なプレーヤー
などの企業メドトロニック (米国)、ボストン サイエンティフィック(米国)、アボット ラボラトリーズ(米国)、バイエル AG(ドイツ)、フィリップス(オランダ)、テルモ コーポレーション(日本)、カーディナル ヘルス(米国)、クック メディカル(米国)、ストライカー コーポレーション(米国)などは、世界市場の主要な参加企業です。