Market Analysis
In-depth Analysis of Recombinant Vaccines Market Industry Landscape
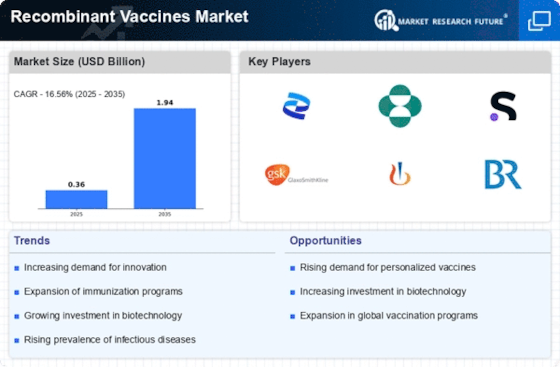
The market dynamics of recombinant vaccines represent a significant aspect of the broader healthcare and biotechnology landscape. Recombinant vaccines, developed through genetic engineering techniques, have emerged as pivotal tools in preventing various infectious diseases. The market is shaped by a multitude of factors, including disease prevalence, technological advancements, regulatory considerations, and global vaccination initiatives.
On influencing the recombinant vaccines market, the prevalence of infectious diseases plays a central role. These vaccines target specific pathogens hence demand is largely influenced by disease epidemiology such as hepatitis B virus (HBV), human papillomavirus (HPV) and influenza. Infections are dynamic entities evolving with time which calls for consistent research and development efforts towards making appropriate vaccines that are proactive to public health needs.
Technological advances have been found to be among the significant determinants of the market dynamics for recombinant vaccines in the industry. Precise manipulation of microbial genes can now be made possible through genetic engineering techniques leading to safer and more effective vaccines. This process improves vaccine development while at the same time affecting manufacturing processes resulting into increased scalability and cost-effectiveness. Market embraces new technologies that improve safety profiles and overall efficacy of recombinant vaccines.
Regulatory matters are critical when determining how this segment behaves within the vaccine’s market structure. Regulation agencies take control over quality assurance procedures before allowing their commercialization thus ensuring their effectiveness and safety aspects are achieved during production processes. Vaccines development standards as well as monitoring after marketing activities need appropriate guidelines from these bodies so that they can continue functioning well in future times. Accordingly, companies who navigate these processes successfully bring on board novel recombinant vaccines which attract interest from consumers.
Global vaccination initiatives contribute to changing markets by creating awareness and increasing accessibility to new products or services such as those offered by Gavi and World Health Organization (WHO). Such programs have often targeted diseases preventable through use of genetically engineered organisms thereby promoting wide spread utilization by national immunization schemes. Considering this, the market evolves to meet new demand arising from increased vaccination programs and further explores new frontiers in terms of collaborations as well as alternative channels for distribution.
Economic issues including healthcare budgets and affordability also have a direct bearing on the recombinant vaccines segment. The cost of research, development, and manufacturing, along with price setting for the vaccines, determine how accessible it will be. There should be affordable immunizations that are still good business sense. Thus, pricing strategies plus reimbursement policies underpinning successful commercialization of recombinant vaccines become part of market dynamics.
Biotechnology and pharmaceutical sectors’ synergies influence the market dynamics with respect to recombinant vaccines. Such partnerships may include academic institutions, manufacturers of vaccine products or even public-private partnerships (PPP). By pooling knowledge bases and resources among each other these alliances make it possible to accelerate vaccine development while at the same time bringing about knowledge sharing within the industry itself. As a result, market trends are influenced by collaborative efforts promoting innovation in response to complex challenges associated with vaccination campaigns targeting various populations across different geographical areas leading to global outbreaks.


















Leave a Comment