契約研究機関市場 概要
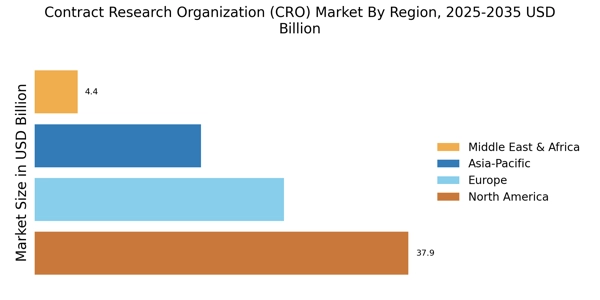
市場調査未来レポートの分析によると、契約研究機関市場の規模は2024年に843億ドルと評価されました。市場は2025年に898.8億ドルから2035年までに1706.3億ドルに成長すると予測されており、2025年から2035年の予測期間中に年平均成長率(CAGR)は6.62%となります。北米は市場をリードしており、44.96%以上のシェアを占め、約379億ドルの収益を生み出しています。
契約研究機関(CRO)市場は、慢性疾患の世界的な負担の増加と効率的な薬剤開発の需要の高まりによって主に推進されています。これにより、製薬会社は臨床研究を専門の組織にアウトソースし、迅速でコスト効果が高く、準拠した試験の実施を促進しています。
世界保健機関(WHO)によると、非感染性疾患は毎年世界の死亡者の74%を占めており(4100万人)、臨床試験の需要が大幅に増加し、CROに対する依存が高まっています。
主要な市場動向とハイライト
契約研究機関市場は、技術革新とアウトソーシングの増加により、堅調な成長を遂げています。
- "北米はCROサービスの最大市場であり、臨床研究に対する堅調な需要を反映しています。
- アジア太平洋地域は、バイオ医薬品活動の拡大により、最も成長が早い市場として浮上しています。
- 臨床研究サービスはCROの風景を支配しており、前臨床サービスは急速に成長しています。
- R&D支出の増加と臨床試験の複雑化が市場拡大を促進する主要な要因です。"
市場規模と予測
| 2024年の市場規模 | 84.3(米ドル十億) |
| 2035年の市場規模 | 170.63(米ドル十億) |
| CAGR(2025 - 2035) | 6.62% |
主要なプレーヤー
IQVIA(米国)、Labcorp Drug Development(米国)、PPD(米国)、Syneos Health(米国)、Charles River Laboratories(米国)、Medpace(米国)、PRA Health Sciences(米国)、Wuxi AppTec(中国)、Celerion(米国)などの企業は、世界市場の主要な参加者の一部です。