分子診断市場 概要
MRFRの分析によると、分子診断市場は2024年に178億米ドルと推定されています。分子診断業界は、2025年に185.9億米ドルから2035年には286.5億米ドルに成長すると予測されており、2025年から2035年の予測期間中に年平均成長率(CAGR)は4.42を示します。
主要な市場動向とハイライト
分子診断市場は、技術の進歩と個別化医療ソリューションへの需要の高まりにより、 substantial growth に向けて準備が整っています。
- 技術の進歩は、特に北米において分子診断の精度と効率を向上させています。腫瘍学セグメントは依然として最大の市場であり、感染症セグメントはアジア太平洋地域で急成長を遂げています。ポイントオブケアテストは注目を集めており、よりアクセスしやすく、迅速な診断ソリューションへのシフトを反映しています。慢性疾患の増加とゲノム技術の進歩は、市場の拡大を促進する重要な要因です。
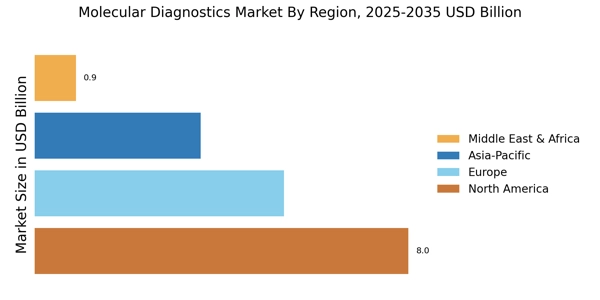
市場規模と予測
| 2024 Market Size | 178億ドル |
| 2035 Market Size | 2865億ドル |
| CAGR (2025 - 2035) | 4.42% |
主要なプレーヤー
ロシュ・ダイアグノスティックス(スイス)、アボット・ラボラトリーズ(アメリカ)、サーモフィッシャー・サイエンティフィック(アメリカ)、シーメンス・ヘルスケア(ドイツ)、ダナハー・コーポレーション(アメリカ)、バイオメリュー(フランス)、キアゲン(オランダ)、ホロジック(アメリカ)、パーキンエルマー(アメリカ)