医療用酸素濃縮器市場 概要
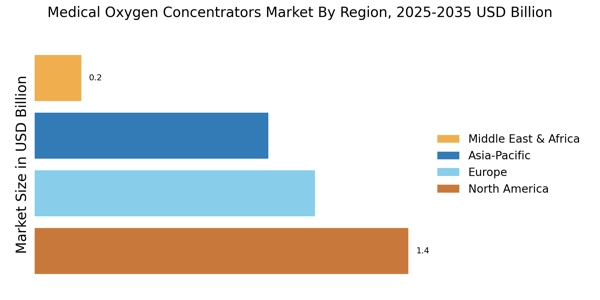
世界の医療用酸素濃縮器市場規模は2024年に35億9,000万米ドルと評価され、市場は2025年の38億1,500万米ドルから2035年までに70億3,000万米ドルに成長すると予測されており、2025年から2035年の予測期間中に6.26%のCAGRを記録します。北米は 2024 年に 40% 以上のシェアで市場をリードし、約 14 億 3,600 万米ドルの収益を生み出しました。
慢性呼吸器疾患の世界的な有病率の上昇と在宅酸素療法の需要の増加が、市場の主要な成長原動力となっています。高齢者人口の拡大と医療へのアクセスの向上により、ポータブルで効率的な酸素供給システムの導入が世界中で加速しています。
IHME (世界疾病負担調査) によると、慢性閉塞性肺疾患 (COPD) は世界中で約 3 億 9,200 万人が罹患しており、長期的な酸素療法の需要が大幅に増加し、医療用酸素濃縮器市場の持続的な成長を支えています。
主要な市場動向とハイライト
医療用酸素濃縮器市場は、技術の進歩と在宅医療ソリューションへの移行によって力強い成長を遂げています。
- 携帯型酸素濃縮器は、機動性、利便性、患者に優しい呼吸補助システムにより、このタイプのセグメントで 63% のシェアを占めています。
- 連続フロー技術は、世界の慢性呼吸器疾患患者に対する信頼性の高い酸素供給によって 66% のシェアを獲得し、リードしています。
- COPDは市場シェアの61%を占めており、これはIHMEが酸素療法を必要とする世界中の約3億9,200万人のCOPD患者を報告していることによって裏付けられています。
- 北米は 1,600 万人の COPD 患者と強力な呼吸器ケアインフラの拡大により、収益シェアが 40% 以上を占めています。
市場規模と予測
| 2024年の市場規模 | 3.59 (USD Billion) |
| 2035年の市場規模 | 7.003 (USD Billion) |
| CAGR (2025 - 2035) | 6.26% |
| 2024 年に最大の地域市場シェアを獲得 | 北米 |