体外診断市場 概要
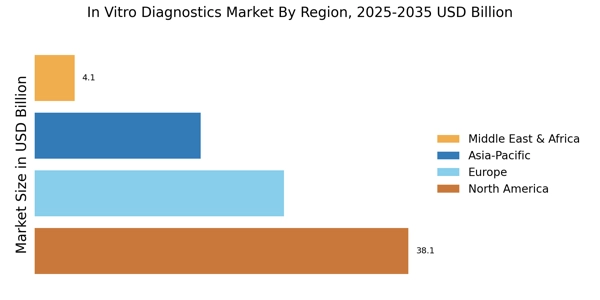
世界の体外診断薬(IVD)市場規模は2024年に846億米ドルと評価され、市場は2025年の881億7000万米ドルから2035年までに1,333億米ドルに成長すると予測されており、2025年から2035年の予測期間中に4.22%のCAGRを記録します。北米は 45% 以上のシェアで市場をリードし、約 380 億米ドルの収益を生み出しました。
慢性疾患の有病率の上昇と疾患の早期発見に対する需要の増加が、体外診断市場の主要な成長原動力となっています。分子診断、AI 対応の検査プラットフォーム、個別化医療の進歩により、診断精度が向上し、治療決定が迅速化され、世界的に患者の転帰が向上しています。
WHO Data Portal によると、慢性疾患による死亡は年間約 4,100 万人に上り、世界の死亡のほぼ 74% を占めています。早期診断、病気のスクリーニング、予防検査に対する医療の関心が高まっているため、世界中の病院、研究所、ポイントオブケアの医療現場で高度な体外診断ソリューションの導入が大幅に加速しています。
主要な市場動向とハイライト
体外診断市場は、技術の進歩とパーソナライズされた医療ソリューションに対する需要の増加によって大幅な成長が見込まれています。
- 北米は 38.1% のシェアを占めており、NIH の 450 億米ドルの資金と年間 10 億件を超える臨床検査に支えられています。
- 欧州は高齢者人口の25%と診断需要の高まりにより、253億8,000万米ドルに達し、シェアは30%となった。
- 個別化医療市場は 2025 年までに 2 兆米ドルに近づき、バイオマーカーベースの体外診断ソリューションの採用が促進されます。
- 予防医療は年間 8% で成長しており、世界中で定期的な診断検査と病気の早期発見が大幅に増加しています。
市場規模と予測
| 2024年の市場規模 | 84.6 (USD Billion) |
| 2035年の市場規模 | 133.3 (USD Billion) |
| CAGR (2025 - 2035) | 4.22% |
| 2024 年に最大の地域市場シェアを獲得 | 北米 |
主要なプレーヤー
などの企業 ロシュ・ダイアグノスティックス(スイス)、アボット ラボラトリーズ (米国)、シーメンス ヘルスニアーズ(ドイツ)、Thermo Fisher Scientific (米国)、Danaher Corporation (米国)、Becton Dickinson and Company (米国)、bioMérieux (フランス)、Ortho Clinical Diagnostics (米国)、Hologic, Inc. (米国) は、世界市場の主要な参加企業です。